Scalable Compliance
Built for Regulated Operations of Any Scale
From single-site operators to global organizations, Seemoto centralizes all monitoring records into a secure, cloud-based audit repository.
- Temperature and condition monitoring records
- Alert and deviation history
- Time-stamped measurements and system events
- Long-term data retention aligned with regulations
No spreadsheets. No paper logs. No gaps.
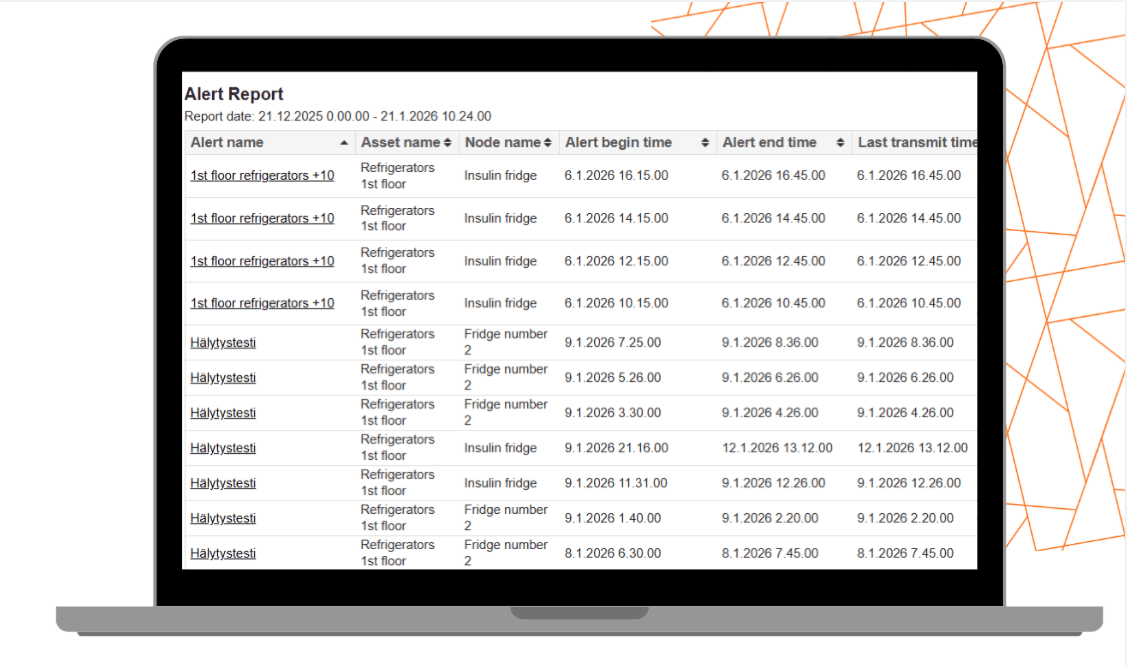
One-Click Reports
Audit-Ready Reports in Seconds
Generate professional audit documentation instantly. Seemoto allows users to:
- Export reports in PDF or CSV
- Select custom time ranges
- Filter by location, sensor, asset, or alarm
- Include min/max/average values, deviations, and timestamps
Reports are structured to support internal audits, customer reviews, and regulatory inspections without additional formatting.
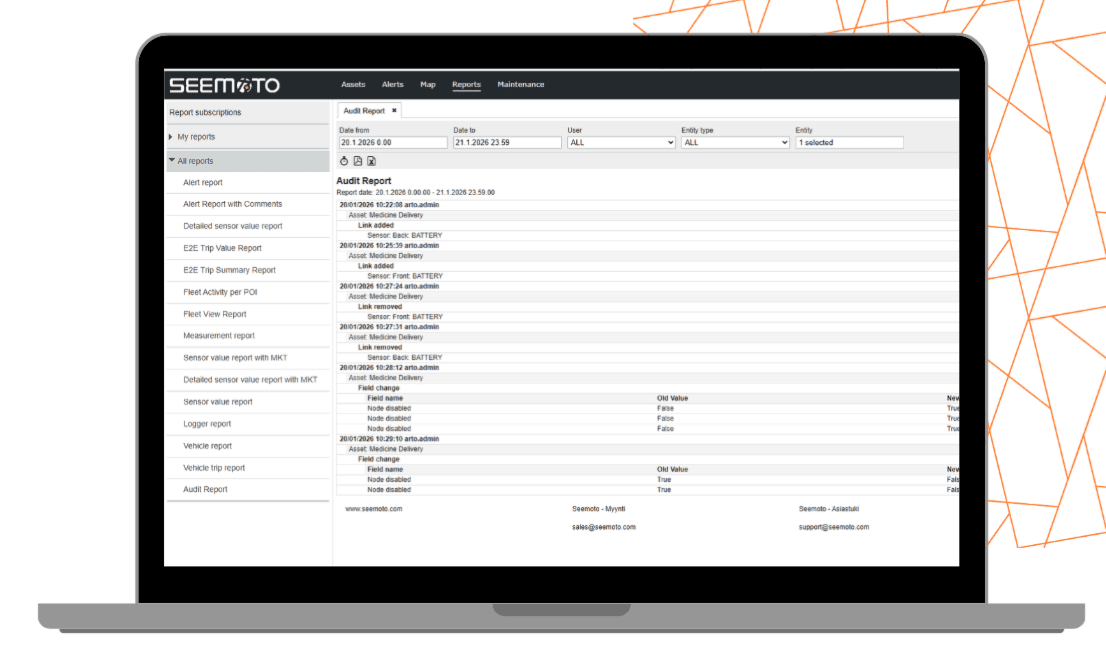
Built-In Compliance Features
Every aspect of the reporting tool is designed to ensure data integrity, traceability, and seamless integration.
Full Traceability & Data Integrity
- Time-stamped
- Tamper-resistant
- Linked to source sensor and location
- Stored securely for up to five years
Aligned with EN12830, GDP, GMP, HACCP, and FDA 21 CFR Part 11 supportive workflows.
Designed to Reduce Audit Risk
- No missing measurements
- No handwritten records
- No manual data transfer
- No inconsistent report formats
Auditors see consistent, chronological, and verifiable records every time.
Seamless Integration
- Real-time monitoring
- Mobile and fixed sensors
- Gateways (GSM/Ethernet)
- Seemoto dashboard and mobile app
No separate systems. No data duplication. Everything flows into a single reporting environment.
Actionable Insights
- Identify recurring deviations
- Compare performance across sites
- Analyze trends over time
- Support CAPA processes
Turn reporting from a compliance burden into a decision-support tool.
Typical Use Cases
From pharma audits to food safety documentation, Seemoto supports compliance across industries.
Pharmaceutical & Healthcare Audits
Temperature compliance reporting for medicines, vaccines, blood, and samples. Full traceability for GDP/GMP inspections.
Food Safety & HACCP Documentation
Automated records for storage, production, and distribution. Digital HACCP logs ready for health inspections.
Warehouse & Logistics Inspections
Proof of compliance across multiple zones and sites. Multi-location reports with one click.
Cold Chain & Transport Validation
Exportable reports for routes, storage periods, and handovers. Complete chain-of-custody documentation.
Internal Quality Reviews
Performance benchmarking across sites and time periods. Data-driven insights for continuous improvement.
Why Seemoto Audit & Reporting
Purpose-built reporting that turns compliance from a burden into a competitive advantage.
Centralized Reporting
All monitoring data from every site flows into one cloud-based reporting environment.
One-Click Exports
Generate professional PDF or CSV reports instantly — no formatting or manual assembly required.
Long-Term Retention
Up to 5 years of secure cloud archiving meets regulatory data retention requirements.
Audit-Ready by Default
Reports are structured for inspections from day one — no preparation needed when auditors arrive.
Reduced Manual Work
Automated logging and reporting eliminates spreadsheets, paper logs, and manual data entry.
Industry Trusted
Used across pharma, food, healthcare, and logistics by organizations that demand compliance confidence.
Frequently Asked Questions
Ready to Simplify Your Reporting?
See how Seemoto's Audit & Reporting Tool can make your operations inspection-ready.
